 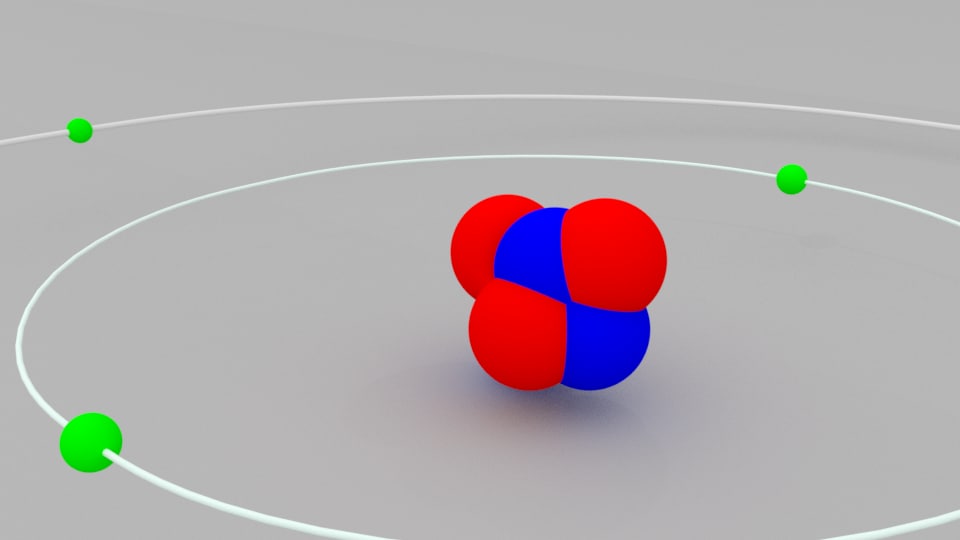
The illustration above uses the hydrogen wavefunctions, which are not exactly correct for lithium but can be used to obtain a qualitative understanding of the dependence of the electron energies on the orbital quantum number. The 2s electron is lowered about 1.7 eV below the n=2 hydrogenic energy level of -3.4 eV which it would have if the shielding were perfect. 
Both levels penetrate enough to be significantly lower than the n=2 hydrogen energy which they would have if the shielding were perfect. The lithium 2s level is significantly lower than the 2p because of greater penetration past the shielding of the 1s electron. Why do levels vary with orbital quantum number? The ionization potential given by NIST was 5.395 eV. Discrepancies may remain with the other levels. The levels up through the 3d plus the 4p and 4d were rescaled to fit the data in the table of neutral lithium levels from NIST. The general layout here was taken from Rohlf but some discrepancies were found in the lower levels. This is true for high angular momentum states as shown, but the s and p states fall well below the corresponding hydrogen energy levels. Since the outer electron looks inward at just one net positive charge, it could be expected to have energy levels close to those of hydrogen. The lithium atom has a closed n=1 shell with two electrons and then one electron outside. As shown in Figure 1 of the Orbitals page, the 2 s electron cloud is bigger than 1 s even in the hydrogen atom with a nuclear charge of only +1.Hydrogen-Like Atoms:Lithium Lithium Energy Levels The first reason is that the principal quantum number n has increased from 1 to 2. There are two reasons why this 2 s cloud is so large. In sharp contrast to this compact inner orbital is the very large and very diffuse cloud of the outer 2 s electron. The superscripts 2 and 1 indicate that there are two electrons in the 1 s orbital and one electron in the 2 s orbital, respectively.Īs in the case of helium, the increase in nuclear charge to +3 produces a corresponding reduction in the size of the lithium 1 s orbital. A convenient shorthand form for indicating this electron configuration is The Pauli principle prevents more than two electrons from occupying this orbital, and so the third (red) electron must occupy the next higher orbital in energy, namely, the 2s orbital. As in the previous atom, two electrons (blue and green) occupy the 1 s orbital. In the dot density image below, the three electrons of the lithium atom are color-coded blue, green, and red. This effect is offset, but to a lesser extent, by the mutual repulsion of the two electron clouds. This pulls both the green and the blue electron clouds in more tightly. This contraction is caused by the increase in the charge on the nucleus from +1 in the hydrogen atom to +2 in the helium atom. The Pauli principle implies that if two electrons have parallel spins, they must occupy different orbitals.Īn obvious feature of the helium atom shown below is that it is somewhat smaller than the hydrogen atom drawn to the same scale above. Two electrons spinning in the same direction are said to have their spins parallel and are indicated by. Two such electrons are said to be spin paired and are often represented by arrows pointing in different directions, i.e., by the symbol. According to the Pauli exclusion principle, if two electrons occupy the same orbital, they must have opposite spins. Two orientations (clockwise and counterclockwise, referred to as spin up or spin down) are possible for this spin. Electrons can not only move about from place to place, but they can also rotate or spin about themselves. This restriction is connected with a property of the electrons not yet discussed, namely, their spin. It turns out that 2 is the maximum number of electrons any orbital can hold. Note that both electrons occupy the same orbital, namely, a 1 s orbital. One electron is color coded in blue, and the other in green. Dot-density diagrams for both these electrons are shown below. The first element in the periodic table with more than one electron is helium, which has two electrons. 
In these diagrams, each electron wave is displayed with a different color and you can toggle the view of each electron wave on and off using the buttons below the dot density diagrams. Thus, more dots indicates more electron density 'cloud' in that region. The images found here depict electron wave density by number of dots. With some familiarity with the properties of single electrons, such as the single electron around the hydrogen nucleus above, we can discuss atoms containing more than one electron. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
